BIOACID II – Consortium 4: Effects of Ocean Acidification in a Warming Climate on Species Interactions at Distribution Boundaries: Mechanisms and Consequences at Ecosystem Level
Lead Proponent: Dr. Felix Christopher Mark (AWI, Bremerhaven)
Ocean acidification is an additional stressor developing in parallel to ongoing climate warming. Future impacts of ocean acidification on organisms and ecosystems are expected to be greatest in Polar Regions, owing to enhanced CO2 solubility in cold waters and body fluids and to the concomitant exposure of organisms to a strong warming trend. At the same time, thermal tolerance windows are narrower and thus sensitivities to combined stressor effects are likely to be higher in cold-adapted polar compared to temperate species. The expected rise in carbon dioxide concentrations and temperature in the oceans (800-1000 μatm and 1-2 °C, respectively, until the year 2100 (IPCC, 2007)) may thus prove to be particularly threatening to Arctic ecosystems.
As the oceans are warming, fish stocks are moving with the water masses of their preferred temperatures to stay within a physiologically optimal temperature range, provided further factors such as food availability and competition with other species allow for that. This has already been documented for several fish species of the North Sea, which have been moving northward at a rate of approximately 12 km per decade (Drinkwater 2009, Perry et al. 2005). In response to this warming trend, the North Arctic stock of Atlantic cod (Gadus morhua) has also shifted spawning areas to the north (Sundby and Nakken 2008) and expanded its range into the Barents Sea (Drinkwater 2009). For the greatest part of the year, juvenile Atlantic cod are now frequently found in the coastal waters of Spitsbergen (Olsen et al. 2010), leading to strong competition with native Polar cod (Boreogadus saida). Polar cod is a dominant fish species on the Arctic shelf and is considered a key species in Arctic ecosystems. It is not clear, however, how these two species will interact in the long term and whether the ecosystem’s species composition will change due to the range expansion of the Atlantic G. morhua into the Arctic (Renaud et al. 2011).
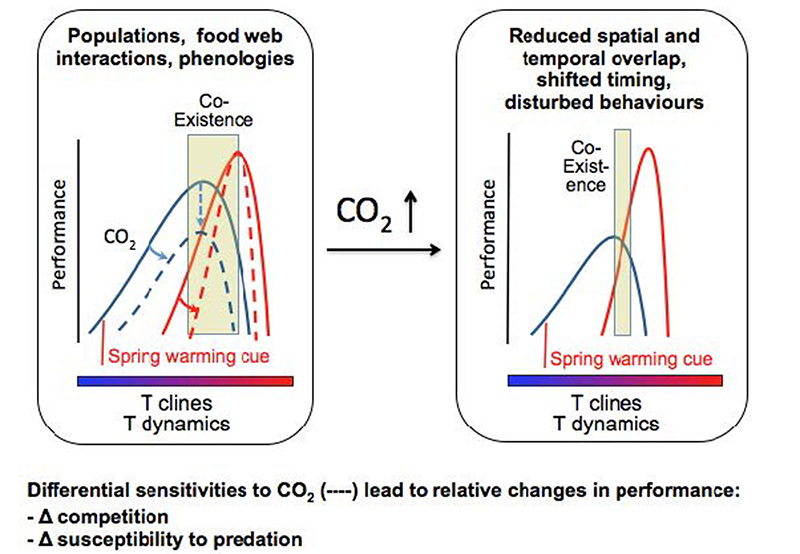
Ecosystem-level perspective of how species interactions are affected by the synergistic effects of shifting temperatures, CO2 and hypoxia levels, building on a mechanistic understanding. The integration of CO2 sensitivity on a thermal matrix as defined by the principles of oxygen- and capacity-limited thermal tolerance emphasizes how species-specific sensitivities and their minimal levels of performance influence the window of temporal and spatial overlap defining the temperature range of coexistence as well as the changes in relative performance. Graph modified from Pörtner and Farrell 2008, Pörtner 2010.
The effects of ocean warming on fish distribution and physiology have been studied and documented to some extent in Atlantic cod (e.g. Colosimo et al. 2003, Lannig et al. 2003, Loeng and Drinkwater 2007, Mueter et al. 2009, Pörtner et al. 2008, Pörtner et al. 2001, Rose 2005) and to a lesser extent in Polar cod (Gjosaeter and Ajiad 1994, Graham and Hop 1995, Hop and Graham 1995, Nahrgang et al. 2010). However, very little is known about how the physiology or distribution of these species is altered by the additional effects of hypercapnia. Moreover, we lack data about how hypercapnia will modify the interactions between species, which are already affected by the warming trend. Using Atlantic cod and Polar cod as model species, we therefore propose to study the competitive interaction between two currently coexisting Arctic fish species under the combined effects of ocean acidification and warming (OAW) in an Arctic ecosystem, the Kongsfjord in Spitsbergen.
In this case study we intend to take into account species physiology, behaviour and life cycle and to investigate the hypothesis that these species will be impacted differently by OAW, with the result of future changes in their competitive interaction. In the coastal waters of Spitsbergen, Atlantic cod find themselves at the northern limit of their geographical distribution and thus at the lower end of their thermal tolerance range. The opposite holds true for Polar cod. Spitsbergen marks the southern boundary of its distribution range and the upper end of its thermal tolerance range. This implies that within the range of their coexistence around Spitsbergen, Atlantic and Polar cod are both energetically limited and their physiological performance should be highly susceptible to further stressors. The increasing concentration of carbon dioxide is clearly one of them (Pörtner 2010). From that perspective, it is our view that ocean acidification may be a critical factor controlling the interaction of coexisting species and that it is likely to determine the outcome of their different abilities to face OAW, i.e. be displaced or restricted to a less favourable habitat (Fig. 4.1, Abrams et al. 2008, Matthews et al. 2010).
For a holistic and integrative analysis of OAW, it is important to consider the different sensitivities and susceptibilities of the various life stages of the two species, as well as their interaction with their prey organisms and thus, their dependence on the food web.
Previous research on the effects of elevated CO2 levels on marine fishes led to the general notion that due to their powerful mechanisms of ion regulation they are not particularly vulnerable to ocean acidification. Adult cod are able to compensate for acid-base disturbances (Larsen et al. 1997) and show no substantial effects of hypercapnia on physiological performance (Melzner et al. 2009a). However, studies in various species demonstrated chronic effects of environmental hypercapnia on juveniles (Moran and Stottrup 2010), and a sensitivity of eggs and early life stages that was higher than in adults. It must be mentioned, however, that all of these observations were reported for CO2 concentrations beyond realistic ocean acidification scenarios (Ishimatsu 2005, Kikkawa et al. 2004). The resulting view that fish are largely insensitive to OA may therefore be premature.
In fact, recent findings demonstrated behavioural disturbances in tropical coral reef fishes exposed to hypercapnia levels according to ocean acidification scenarios (Munday et al. 2009c); as well as alterations in larval development of cod (Frommel et al. in press) and herring (Franke and Clemmesen 2011). Also, the hypothesis that sensitivity to thermal extremes is enhanced under projected ocean acidification levels was confirmed in coral reef fish (Nilsson et al. 2009, Pörtner and Farrell 2008). In general, sensitivity of fishes to ocean acidification may be higher at temperature extremes, a hypothesis to be tested in fishes from various climate zones. At the same time, the allometry of thermal limitation indicates that tolerance to temperature extremes varies across life stages and decreases with increasing body size (e.g. Pörtner et al. 2008, Pörtner and Knust 2007, Storch et al. 2011). Accordingly, sensitivity to OAW may also vary during ontogeny as recently seen in crustaceans (Schiffer et al. in prep). The consequences of these observations for larval recruitment also remain to be explored. This fragmented knowledge base clearly calls for a better understanding of how exposure to combined CO2 and temperature scenarios contribute to shaping fitness windows. This includes the question whether all life stages respond similarly or whether some of them represent potential bottlenecks for population survival.
Most life stages of Atlantic and Polar cod feed on planktonic organisms (Levasseur et al. 1994, Lowry and Frost 1981, Renaud et al. 2011). Their availability and nutritional quality are a very important secondary effect of OAW, likely influencing development and performance of the early life stages of both fish species. Driven by climate change, plankton communities undergo well-documented shifts in composition and spatial ranges (Beaugrand et al. 2009), which, in a bottom-up control process, influences the biogeographic distribution of the fish stocks that prey upon them (Drinkwater 2009, Drinkwater 2006). In this respect, it is of considerable importance, that, by nature, invasive species are more likely to be generalists than specialists and thus might be more successful than natives at adapting to changes in food availability and composition (Dukes and Mooney 1999). In the coastal waters of Spitsbergen, this is mirrored in the diets of Atlantic and Polar cod (Renaud et al. 2011) that show a more varied food composition for Atlantic cod. This might prove advantageous for the fitness and potential of Atlantic cod to adapt to a changing climate.
The success of predation, and thus survival, also depends on dedicated behaviour of the predator and its prey. Studies in tropical coral reef fish revealed that exposure to increased CO2 strongly disturbed avoidance behaviour in juveniles and, as a consequence, resulted in their enhanced predation. These behavioural changes are presumably elicited by hypercapnia effects on the central nervous system. By changing species behaviour, hypercapnia likely has the potential to overthrow the balanced food-web interactions in this particular ecosystem (Ferrari et al. 2011, Munday et al. 2009a, Munday et al. 2010). This aspect has never been studied in adult or juvenile temperate and boreal fish and therefore needs to be addressed in the interactions between Atlantic and Polar cod and their prey.
The Gadidae family comprises some of the commercially most important fish species in the North Atlantic, namely cod, haddock, pollack and whiting, and Polar cod has also been the target of substantial industrial fishery in the Arctic (FAO Fishery Statistic). The Atlantic cod is now the subject of intensive aquaculture in Norway. It is therefore clear that any shift in the population structure, caused by OAW could have far reaching effects not only on the ecosystem itself but also on fisheries. It has also been suggested that OAW may have deleterious effects on aquaculture (Guinotte and Fabry 2008). The socio–economic consequences of such scenarios need to be explored.
General objectives:
This consortium sets out to investigate how the combined effects of OAW will affect different life stages of the interacting fish species Gadus morhua and Boreogadus saida and their prey. Its strength lies in the integrative approach across levels of biological organisation, from the genome to the ecosystem. Tightly intertwined work packages, which will rely on several joint acclimation experiments, will allow a thorough analysis of putatively shifting species interactions and their implications at ecosystem and socioeconomic levels.
Objectives include addressing the question whether OAW affects interacting species differently due to divergent physiological optima and ranges, expressed in thermal tolerance windows and associated performance capacities and phenologies of specific life stages. Crucial mechanisms as well as causes and effects will become accessible from unravelling the connections between levels of biological organisation, from genomic, molecular to cellular, individual and population level. Scopes for acclimation (physiology and behaviour) and adaptation (evolution) that together define species resilience will be studied in various life stages (eggs, larvae, juveniles, adults) and the most sensitive one(s) identified. Functional determinants of individual fitness such as ion and acid-base regulation, mitochondrial energy metabolism and immune response will also be examined. Furthermore, it will be addressed how these processes as well as fitness may be influenced by food quality and availability.
This consortium will address the following questions:
- How will OAW influence species distribution, competitiveness and behaviour (WP 4.1 and 4.6)?
- Will food web mediated OAW effects influence the vulnerability of species (WP 4.5, 4.8, 4.9)?
- Which life stages are most susceptible to OAW and contribute most to ecosystem level effects (WP 4.1, 4.4, 4.5, 4.6 and 4.7)?
- How do OAW effects become manifest on different levels of biological organisation, how are they linked and interdependent across levels of organisation (WP 4.1, 4.2, 4.3, 4.4, 4.5 and 4.7)?
Research approaches:
Each of the nine work packages in this consortium will engage with a specific level of biological organisation to have their results integrated into a projection of responses at ecosystem level and their socioeconomic consequences in consortium 5 (WP 5.6).
Experiments will be conducted at various laboratories. At the Alfred Wegener Institute in Bremerhaven, Germany, adult and juvenile fish as well as copepods will be kept in specifically designed OAW acclimation systems that were refined (and in part already acquired) during BIOACID I. Cultures of larval and juvenile fish and pteropods will be set up under laboratory conditions at the University of Bergen, Norway and of juvenile fish, pteropods and copepods at the AWIPEV laboratory in Ny-Alesund, Spitsbergen. Fish and copepods will be caught from wild populations with RV Heincke around Spitsbergen and Norway in 2012 and 2013 in close cooperation with our Norwegian project partners in Bergen and Tromsø. We will further include fish and eggs from Norwegian aquaculture and also rely on various life stages cultured at the Sven Lovén Centre for Marine Sciences in Kristineberg, Sweden (Atlantic cod adults, eggs, larvae) and the National Cod Breeding Centre in Tromsø (Polar cod adults, eggs, larvae).
Work packages 4.1, 4.2, 4.3, 4.6 and 4.7 will rely on juvenile animals from common incubations of juvenile Atlantic cod (spring 2013) and Polar cod (spring 2014) at various temperature-pCO2 combinations at the Alfred Wegener Institute. These incubations (organized by WP 4.1) will thus serve as a central hub for several work packages, ensuring thorough connections between the work packages while at the same time reducing the effort and number of individuals needed per incubation.
To trace individual acclimation histories for each juvenile fish, individuals from all stocks will be tagged and populations screened for their bandwidth of growth and other environmental adaptation-relevant physiological performances (‘whole organism phenotype’, WP 4.1), as well as for genetic variability (‘genotypes’, WP 4.3). The studies will also define the diversity of responses from which selection and further adaptation will be possible, increasing the resolution and accuracy for predictions at the population level. Analysis of transcriptomic responses (WP 4.3) along with adaptations of the proteome (WP 4.2) will provide information on adaptation mechanisms, which become manifest at cellular and systemic levels.
Consideration of within population diversity will increase the resolution and accuracy for predictions at the population level. Such predictions also require the identification of the most sensitive life stages and transition phases, including reproduction, fertilisation success, egg and larval development and larvae to juvenile metamorphosis (WP 4.4 and 4.5). Species-specific differences and shifts in performance characters are hypothesized to shape interactions within food webs (WP 4.5 and 4.6). CO2 may affect such processes due to changes in relative performance and in behaviour. WP 4.6 will set out to identify behavioural changes in juvenile fish and elucidate the neuronal underpinning of OAW effects on behaviour using MRI and NMR spectroscopy.
For an inclusion of food web effects, we will integrate the copepods and pteropods reared under the same schemes of OAW in WP 4.8 and WP 4.9 and quantify prey uptake, catch rate by juvenile cod (WP 4.6) as well as potential changes in their food quality (WP 4.8 and 4.9).
To this end, work packages 4.8, 4.9, 4.4, 4.5 and 4.7 will conduct joint experiments on copepod and pteropod performance, on the responses of eggs and fish larvae to CO2 and changing food availability, one to be carried out in summer 2013 at AWIPEV in Ny-Alesund, in spring/summer 2014, Bergen, Norway, and at the AWI facilities. Experiments will be carried out in close collaboration with our external co-operators. For testing the role of such phenomena under field conditions, these work packages will also join BIOACID consortium 1 during the common mesocosm studies (KOSMOS) in Gullmar Fjord, Kristineberg, Sweden, in spring and summer 2013.
In cooperation with the bridging WP 5.6 in consortium 5, we will finally address the socio- economic implications for fishery and aquaculture and establish their societal relevance. To do so, we will connect to an international network of ocean acidification researchers who already investigate the socioeconomic aspects of OAW. This will provide a means to further extrapolate from the findings of this consortium and disseminate them to the public.