The pH
The pH indicates how acidic or basic (alkaline) a liquid is. It depends on the concentration of hydrogen ions in an aqueous solution. If the number of hydrogen ions decreases, the pH increases. If the number of hydrogen ions increases, the pH decreases.
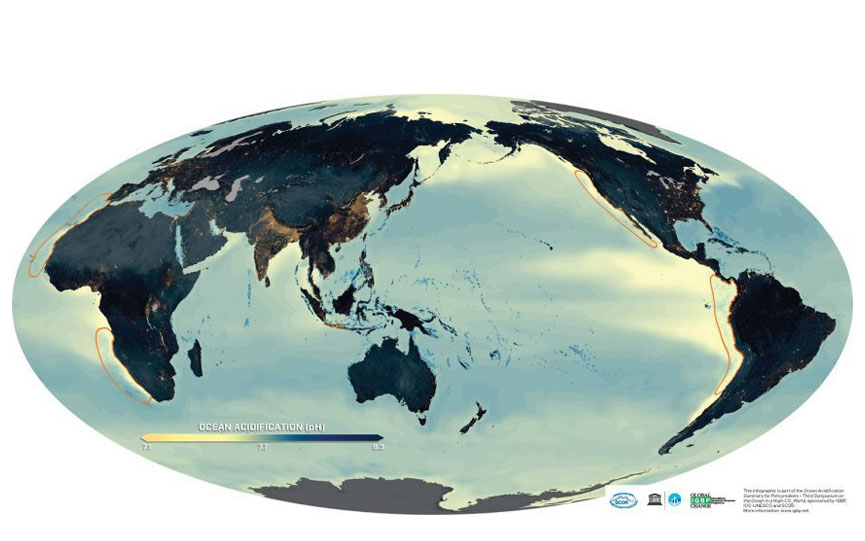
Pure water has a pH of 7. Lower values are described as acidic, and higher ones as basic. Since the pH is based on a logarithmic scale, a change from pH 8 to pH 7 corresponds to a tenfold increase in acidity. If the pH of a liquid changes from 7 to 6, there are ten times as many additional hydrogen ions as in a change from pH 8 to pH 7.
Since the beginning of the Industrial Revolution, the average pH of the global ocean surface has already fallen from 8.2 to 8.1, corresponding to an increase in acidity of about 26 per cent. Values of 7.8 to 7.9 are expected by 2100, representing a doubling of acidity compared to the time before the industrial revolution.
It is unlikely that the open-ocean surface layer will ever become truly acidic (drop below pH 7.0), because seawater is buffered by dissolved salts. That’s why scientists emphasize it “acidifies” or becomes “more acidic”, but not “acidic”. The term “ocean acidification” refers to a pH shift towards the acidic end of the pH scale – similar to the way we would describe an increase in air temperature from -20 degrees Celsius to 0 degrees Celsius: The air is still cold, but we say “it is “warming.”